Case Study: Choosing a Distribution Model for the Chinese Market
By Zhou Qian and Matthew Zito
When investing in China’s medical device industry, especially for those seeking to operate as medical device trading companies, one big question is how to make suitable arrangements for product distribution in China. Based on the diverging circumstances and objectives of investors, there is no one-size-fits-all model. Familiarity with the pros and cons of differing models can help you make the right choice for your business in China. To illustrate these, we have designed the following case study.
Scenario:
V is a medical device company registered in Europe, manufacturing wound care products and orthopedic implants. The company has conducted thorough market research and after careful consideration, decided to invest in China’s medical device industry. V has already registered a subsidiary in Guangzhou, China. (“V-FICE”), which has been set up as a foreign-invested commercial enterprise (FICE) and obtained all necessary licenses for operating in China.
V and V-FICE are now in the process of assessing the best possible model for conducting their import business. At the same time, they are considering whether to go it alone or contract the services of a local Chinese distributor, C, having good credit and connections in the medical device industry in South China.
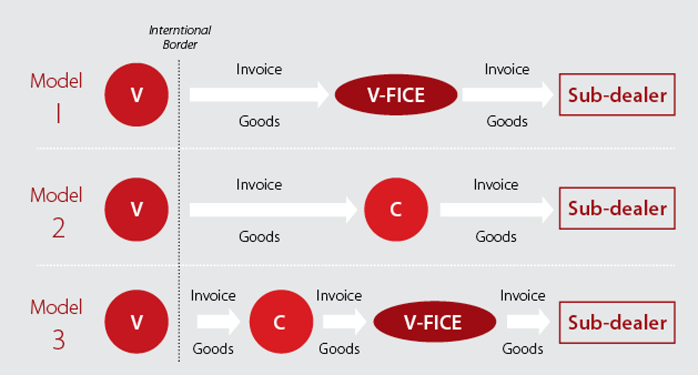
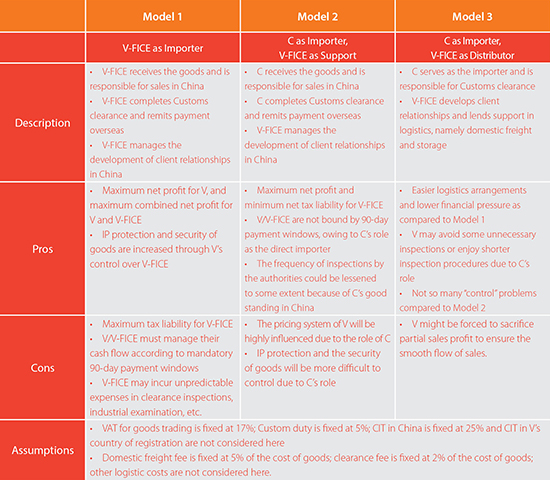
To analyze the different models of distribution available to V, we present the following chart, taking into account the pros and cons of each model in terms of their relevant corporate structures, costs, tax liability, Customs procedures and IP Risk.
Conclusion
As China’s population ages and its living standards improve, the demand for medical devices is only set to increase. Foreign companies looking to enter the Chinese market are well-advised to carefully consider the advantages of different distribution models.With our specialized knowledge and experience in the medical industry, Dezan Shira & Associates can help you to newly establish or grow your operations in China and beyond.
 This article is an excerpt from the November issue of China Briefing Magazine, titled “China Investment Roadmap: The Medical Device Industry.” In this issue of China Briefing, we present a roadmap for investing in China’s medical device industry, from initial market research, to establishing a manufacturing or trading company in China, to obtaining the licenses needed to make or distribute your products. This article is an excerpt from the November issue of China Briefing Magazine, titled “China Investment Roadmap: The Medical Device Industry.” In this issue of China Briefing, we present a roadmap for investing in China’s medical device industry, from initial market research, to establishing a manufacturing or trading company in China, to obtaining the licenses needed to make or distribute your products. |
![]()
 China Retail Industry Report 2014
China Retail Industry Report 2014
In this special edition of China Briefing, we provide an overview of the retail industry in China and the procedures for setting up a retail shop, focusing specifically on brick-and-mortar physical retail stores. Further, we have invited our partner Direct HR to offer some insights on the talent landscape in the retail industry, as well as tips for recruiting retail personnel in China.
 Adapting Your China WFOE to Service China’s Consumers
Adapting Your China WFOE to Service China’s Consumers
In this issue of China Briefing Magazine, we look at the challenges posed to manufacturers amidst China’s rising labor costs and stricter environmental regulations. Manufacturing WFOEs in China should adapt by expanding their business scope to include distribution and determine suitable supply chain solutions. In this regard, we will take a look at the opportunities in China’s domestic consumer market and forecast the sectors that are set to boom in the coming years.
 Revisiting the Shanghai Free Trade Zone: A Year of Reforms
Revisiting the Shanghai Free Trade Zone: A Year of Reforms
In this issue of China Briefing, we revisit the Shanghai FTZ and its preferential environment for foreign investment. In the first three articles, we highlight the many changes that have been introduced in the Zone’s first year of operations, including the 2014 Revised Negative List, as well as new measures relating to alternative dispute resolution, cash pooling, and logistics. Lastly, we include a case study of a foreign company successfully utilizing the Shanghai FTZ to access the Outbound Tourism Industry.
- Previous Article China Announces Three New Free Trade Zones in Tianjin, Guangdong and Fujian
- Next Article “Nali Lai de?” – An Overview of Expat Demographics in China