New Measures to Support Hong Kong and Macao Medical Institutions in Qianhai Cooperation Zone
The Shenzhen Qianhai Administration has proposed new measures to support the establishment and work of Hong Kong and Macao medical institutions within the Qianhai Cooperation Zone. We look at the highlights, benefits, and requirements presented in the provisions.
On July 3, 2023, the Shenzhen Qianhai Administration released the draft Measures of Qianhai Shenzhen-Hong Kong Modern Service Industry Cooperation Zone on Supporting the Agglomeration Development of Hong Kong and Macao Medical Institutions for public comments.
The Shenzhen Qianhai Administration recognized that while there is a considerable number of residents and employees from Hong Kong, Macao, and the international community residing within the Qianhai Cooperation Zone, the limited presence of Hong Kong and Macao medical institutions fails to effectively meet the demand for medical services, and thus, falls short of the zone’s positioning as a modern service industry cooperation zone.
In this article, we will delve into the details of the draft Measures Supporting the Agglomeration Development of Hong Kong and Macao Medical Institutions, the comprehensive measures proposed to support the development and growth of medical institutions within the Qianhai Cooperation Zone, and the eligibility criteria and application requirements for interested entities.
What is the Qianhai Cooperation Zone?
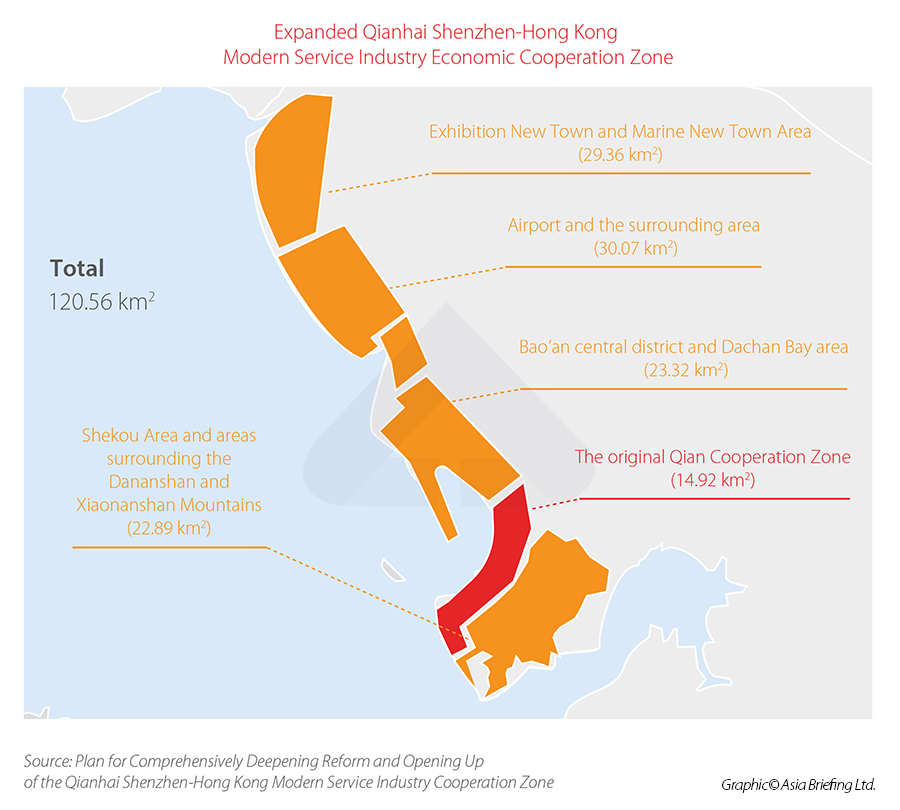
The Qianhai Shenzhen-Hong Kong Modern Service Industry Economic Cooperation Zone (hereinafter, ‘Qianhai Cooperation Zone’), was initially established on August 26, 2010, with the official approval of the State Council. Its primary objective was to serve as an experimental business zone, facilitating enhanced collaboration and interaction between Mainland China and Hong Kong in key sectors, such as finance, logistics, and IT services.
On September 6, 2021, China officially announced the expansion of the zone, whose area was increased by eight times – from 15 sq km to 120 sq km – and has since become pivotal in facilitating cross-border collaboration across various industries.
With its expansion and the incorporation of the Shekou Area, the Qianhai Cooperation Zone is set to become a dynamic hub for economic integration between Mainland China, Hong Kong, and Macao.
This integration allows for the sharing of expertise, resources, and innovation, thereby fostering synergies that can drive growth and development. For example, the Qianhai Cooperation Zone taps into the strengths of Hong Kong and Macao in areas like artificial intelligence (AI), healthcare, fintech, smart cities, the Internet of Things (IoT), and new energy materials.
New measures to support Hong Kong and Macao medical institutions
The draft Measures, which consist of five chapters and 26 provisions, outline a set of comprehensive measures aimed at supporting the establishment and growth of medical institutions in the Qianhai Shenzhen-Hong Kong Modern Service Industry Economic Cooperation Zone (hereinafter ‘Qianhai Cooperation Zone’).
The draft Measures primarily focus on providing support to hospitals, outpatient departments, and clinics established by service providers from Hong Kong and Macao in the zone. Additionally, the document includes several initiatives aimed at supporting medical equipment purchases for relevant institutions, as well as aiding in areas such as medical venues, practice, insurance, and hospital rating support.
Basic financial support for eligible medical institutions in the Qianhai Cooperation Zone
Eligible medical institutions from Hong Kong and Macao can apply for financial support offered by the Qianhai government. These subsidies will be disbursed over a period of three years, with an annual distribution rate of 50 percent, 30 percent, and 20 percent, respectively.
Specific amounts available for support vary based on the institution types, as described in the table.
| Subsidies for Hong Kong and Macao Medical Institutions | |
| Type of legal entity | Available subsidies |
| General hospital wholly owned by Hong Kong and Macao service providers |
|
| General hospital jointly owned by Hong Kong and Macao service providers and Chinese mainland service providers |
|
| Specialized hospital wholly owned by Hong Kong and Macao service providers |
|
| Specialized hospital jointly owned by Hong Kong and Macao service providers and Chinese mainland service providers |
|
| Outpatient department wholly or jointly established by Hong Kong and Macao service providers |
|
| Medical clinics wholly or jointly established by Hong Kong and Macao service providers |
|
Other subsidies
The draft Measures outline various categories of subsidies available for eligible Hong Kong and Macao medical institutions, as summarized in the table below:
| Subsidies for Hong Kong and Macao Medical Institutions |
||
| Subsidy type | Description |
Special conditions and/or limitations |
| Medical equipment procurement | Hong Kong and Macao medical institutions that intend to purchase individual or sets of medical equipment exceeding RMB 500,000 (US$70,068) can apply to receive a subsidy. | This subsidy is designed to assist with the procurement costs and is capped at 10 percent of the actual price of the equipment.
Additionally, it is important to note that the subsidy provided for a single piece of equipment should not exceed RMB 500,000 (US$70,068), while the total subsidy granted to a single institution must not surpass RMB 5 million (US$700,682). |
| Acquisition or rental of medical facilities |
|
|
| Hiring specialized personnel | Hong Kong and Macao medical institutions that hire medical professionals Hong Kong and Macao, and successfully complete their registration in Mainland China, are eligible to a one-time subsidy of RMB 20,000 (US$2,802) per person.
Additionally, when hiring doctors from Hong Kong and Macao that provide substantial diagnosis and specialized treatment services, the medical institutions are eligible to apply for a subsidy of RMB 100 (US$13.96) per visit. |
The maximum annual subsidy available for this category is RMB 1 million (US$140,136) per year, while limit of the cumulative subsidy per institution is RMB 3 million (US$ 420,409) in total. If the institutions have already received some types of subsidies for medical services from the Shenzhen municipal government, they are not eligible for the above-mentioned ones. |
| Insurance for personnel | Eligible medical institutions can receive a cashback equal to 50 percent of the annual premium purchased for their doctors. | The maximum subsidy amount available per year is set at RMB 200,000 (US$20,802). |
| Hospital accreditation | Hong Kong and Macao medical institutions that achieve the “Hospital Quality International Certification Standard” for the first time through evaluation by the Shenzhen Municipal Health Commission Hospital Evaluation and Evaluation Research Center are eligible for a one-time reward of RMB 2 million (US$ 280,272) | N/A |
| “Hong Kong and Macao Medicine and Medical Device Port” | Designated medical institutions under the “Hong Kong and Macao Medicine and Medical Device Port” policy can apply for a one-time reward of RMB 500,000 (US$70,068). Additionally, institutions approved to use urgently needed imported medicines or medical devices from Hong Kong and Macao can receive one-time rewards of RMB 50,000 (US$7,068) for first-time applications and RMB 20,000 (US$2,802) for subsequent applications. | The maximum annual reward for a single institution is RMB 1 million (US$140,136), and the cumulative maximum reward within the policy’s duration is RMB 3 million (US$ 420,409). |
| International commercial insurance | Hong Kong and Macao medical institutions that collaborate with insurance institutions or third-party organizations to provide direct settlement services for cross-border medical insurance claims can apply for a subsidy of RMB 50 (US$7.01) per claim. | The maximum annual subsidy for a single institution is RMB 500,000 (US$70,068), and the cumulative maximum subsidy within the policy’s duration is RMB 1 million (US$140,136). |
| Key specialties and disciplines | Hong Kong and Macao medical institutions participating in national, Guangdong Province, or Shenzhen municipal medical projects related to key specialties or distinctive traditional Chinese medicine specialties can apply for one-time rewards.
The rewards range from RMB 1 million (US$140,136), RMB 500,000 (US$70,068), to RMB 200,000 (US$20,802), depending on the project’s nature. |
This is a one-time support. |
| Key projects | Key projects in the Qianhai Cooperation Zone that contribute significantly to cooperation among Shenzhen, Hong Kong, and Macau, as well as the development of medical services in the region, may enter into separate cooperation agreements with the Qianhai Administration Bureau. | The total number of supported institutions through this approach should not exceed 10 percent of the total number of supported institutions in a given year, and the allocated funds should not exceed 25 percent of the total subsidy amount. |
Which medical institutions are eligible to receive the funds?
The proposed measures apply to all Hong Kong and Macao medical institutions established as wholly owned or joint venture entities in Qianhai Cooperation Zone.
Other eligibility criteria include:
- The medical institutions’ registration, operation, and tax payment must be within the Qianhai Cooperation Zone.
- Must have completed medical institution registration and be registered as Hong Kong, Macao, or Taiwan-invested enterprises.
- They must not have medical accidents or violations in the past year.
It is important to note that specific requirements and conditions may apply to each form of financial support, and additional details may be available in the respective articles or regulations.
What is the application process?
To be eligible for the support funds, Hong Kong and Macao medical institutions need to meet certain application requirements. The application process is outlined in Chapter 3 of the regulations.
Key steps of the application procedure include:
- Eligibility: Only Hong Kong and Macao medical institutions that comply with the regulations and operate within the Qianhai Cooperation Zone are eligible to apply for the support funds. These institutions should meet the specified criteria and have the necessary licenses and permits to provide medical services.
- Application period: The Qianhai Administration Bureau will announce the designated period for accepting applications. Eligible institutions should submit their application materials within this specified timeframe.
- Application materials: The application materials must be prepared and submitted according to the guidelines provided by the Qianhai Administration Bureau.
- Document review and on-site inspection: The Qianhai Administration Bureau reviews the submitted application materials to assess the eligibility and compliance of the institution. If the documents pass the initial review, the Bureau conducts an on-site inspection to further verify the institution’s suitability for receiving the support funds.
- Public announcement and objection period: Institutions that successfully complete the on-site inspection and meet the requirements undergo a public announcement process by the Qianhai Administration Bureau. The public notice period lasts for a minimum of five working days. During this period, any organization or individual can raise objections to the announced institutions.
- Fund disbursement: After the public announcement period, the Qianhai Administration Bureau reviews any objections raised. If there are no valid objections or the objections are found to be invalid, the Bureau proceeds with the disbursement of the corresponding support funds, which are allocated according to the provisions.
What are the required materials?
Specific application materials will include:
- Application form.
- Copy of the business license.
- Copy of the medical institution’s practicing license.
- Declaration of no violations or records of dishonesty.
- Copy of the identification card of the legal representative.
- Materials related to the operating premises in the Qianhai Cooperation Zone.
- Other materials such as relevant qualifications, certifications, and tax payment certificates obtained in the past that can assist the Qianhai Administration Bureau in verifying the application qualifications.
Summary
All in all, the recently published measures by the Shenzhen Qianhai Administration aim to support the integrated development of the Qianhai Cooperation Zone, including the necessary advancement of its healthcare facilities and allied infrastructure.
The proposed financial support, including subsidies for medical equipment purchase, acquisition or rental of medical facilities, hiring specialized personnel, insurance support, hospital accreditation, etc. will speed up the progress of implementation, attract fresh investments, and boost the zone’s appeal to multinational entities seeking to set up in a business-friendly zone with cross-border relaxations.
The draft measures, overall, emphasize the Qianhai Cooperation Zone’s commitment to promoting collaboration among Mainland China, Hong Kong, and Macao regions. These efforts are expected to have a positive impact on the Greater Bay Area by facilitating cross-border integration of industries and services.
About Us
China Briefing is written and produced by Dezan Shira & Associates. The practice assists foreign investors into China and has done so since 1992 through offices in Beijing, Tianjin, Dalian, Qingdao, Shanghai, Hangzhou, Ningbo, Suzhou, Guangzhou, Dongguan, Zhongshan, Shenzhen, and Hong Kong. Please contact the firm for assistance in China at china@dezshira.com.
Dezan Shira & Associates has offices in Vietnam, Indonesia, Singapore, United States, Germany, Italy, India, and Russia, in addition to our trade research facilities along the Belt & Road Initiative. We also have partner firms assisting foreign investors in The Philippines, Malaysia, Thailand, Bangladesh.
- Previous Article Guangzhou’s Talent Green Card System: Benefits, Eligibility, and Application Process
- Next Article Business Opportunities for Foreign Brands in the Children’s Snacks Industry in China