Investing in China’s Medical Device Industry, Part 1
 By Zhou Qian, Kyle Freeman and Matthew Zito
By Zhou Qian, Kyle Freeman and Matthew Zito
Investing in China’s medical device industry requires knowledge of several key regulations governing the industry, including China’s complex and idiosyncratic licensing scheme for medical devices. In this article, we highlight several of these, as well as provide the most up-to-date information regarding the medical device industry’s place in China’s Catalogue of Industries for Guiding Foreign Investment.
Regulations on the Supervision and Administration of Medical Devices (State Council Order No. 650), effective June 1, 2014
Order 650 is the highest level regulation governing the registration and record-keeping of medical devices, requirements for product manufacturing and distribution, and liability for illegal behavior. A revised version of the Regulations on the Supervision and Administration of Medical Devices (2000), the 2014 amendment introduces the following reforms of the regulatory regime for medical devices in China:
- Improvement to the risk-based classification system of medical devices
- Revocation of the re-registration requirements
- Changed order of approval to encourage innovation
- Heavier punishment for violations
- Stronger after-sales supervision
China Food and Drug Administration (CFDA) regulations
To support the implementation of Order No. 650, the China Food and Drug Administration (CFDA) has passed several legal regulations in the past year regarding the registration, manufacture and distribution of medical devices, including:
- Administrative Measures for the Registration of Medical Devices (CFDA Order No. 4)
- Administrative Measures for the Registration of In Vitro Diagnostic Reagents (CFDA Order No. 5)
- Administrative Rules for the Instructions and Labelling of Medical Devices (CFDA Order No. 6)
- Administrative Measures for the Supervision of Medical Device Manufacturing (CFDA Order No. 7)
- Administrative Measures for the Supervision of the Distribution of Medical Devices (CFDA Order No. 8)
![]() RELATED: Market Overview: The Medical Device Industry in China
RELATED: Market Overview: The Medical Device Industry in China
Catalogue of Industries for Guiding Foreign Investment (2011 Revision)
The Catalogue is the framework document for setting restrictions/prohibitions on foreign investment into certain industries in China, and is jointly issued by the National Development and Reform Commission and Ministry of Commerce. Regarding the medical device industry specifically, the revised version of the Catalogue from 2011 removed previous restrictions on the manufacture of non-auto-disposable syringes, infusion sets, blood transfusion devices and blood bags – giving foreign investors access to the manufacture and distribution of all types of medical devices in China.
The Catalogue also includes several types of high-end devices under the category of industries “encouraged” for foreign investment, including electronic endoscopes, fundus cameras, key components of medical imaging equipment, (3D) ultrasonic transducers for medical use, equipment for boron neutron capture therapy (BNCT), image-guided intensity modulated radiation therapy systems, hemodialysis machines and hemofiltration machines, as well as equipment for fully automated enzyme immunoassay (EIA) systems.
Most recently, further revisions to the Catalogue were released on Nov. 4, 2014 in the form of an opinion-seeking draft (the deadlined for public commentary is Dec. 3, 2014). In its current draft, the revised Catalogue newly includes several types of devices under the Encouraged category, namely, blood cells analyzers, automatic chemiluminescence immune analyzers, and high throughput sequencing systems.
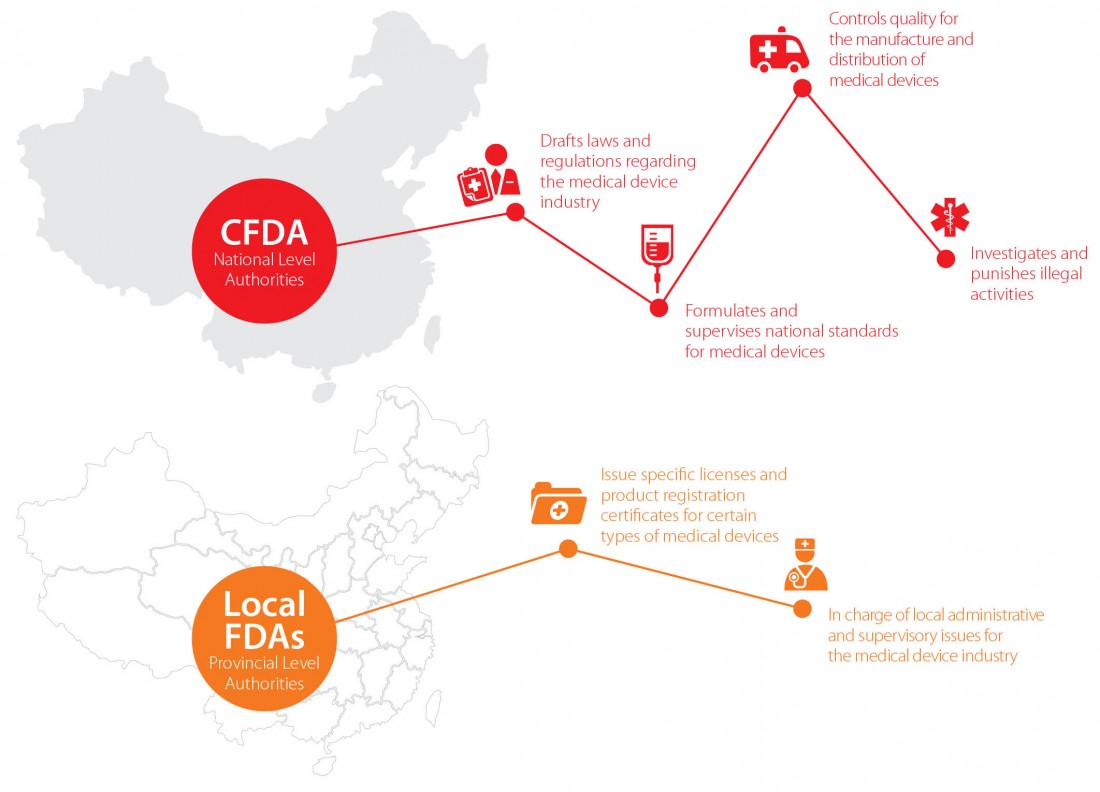
Supervisory and administrative authorities for the medical device industry
In China, the supervisory and administrative authorities for the medical device industry can be classified at the national or local level. At the national level, the China Food and Drug Administration (“CFDA”), known as the State Food and Drug Administration (“SFDA”) prior to 2013, is in charge of the regulation and control of the medical device industry for the whole of China.
In practice, the CFDA delegates a great deal of administrative powers to its local branches, the Food and Drug Administrations (“FDAs”) of provinces, autonomous regions and directly-controlled municipalities.
These bodies are authorized to issue specific licenses and product registration certificates to medical device companies at the provincial level. One thing worth noting for foreign investors, however, is that local FDAs may have more detailed and specific examination and approval requirements than those of the CFDA. Consequently, when investing in the medical device industry, both national and local requirements must be adhered to.
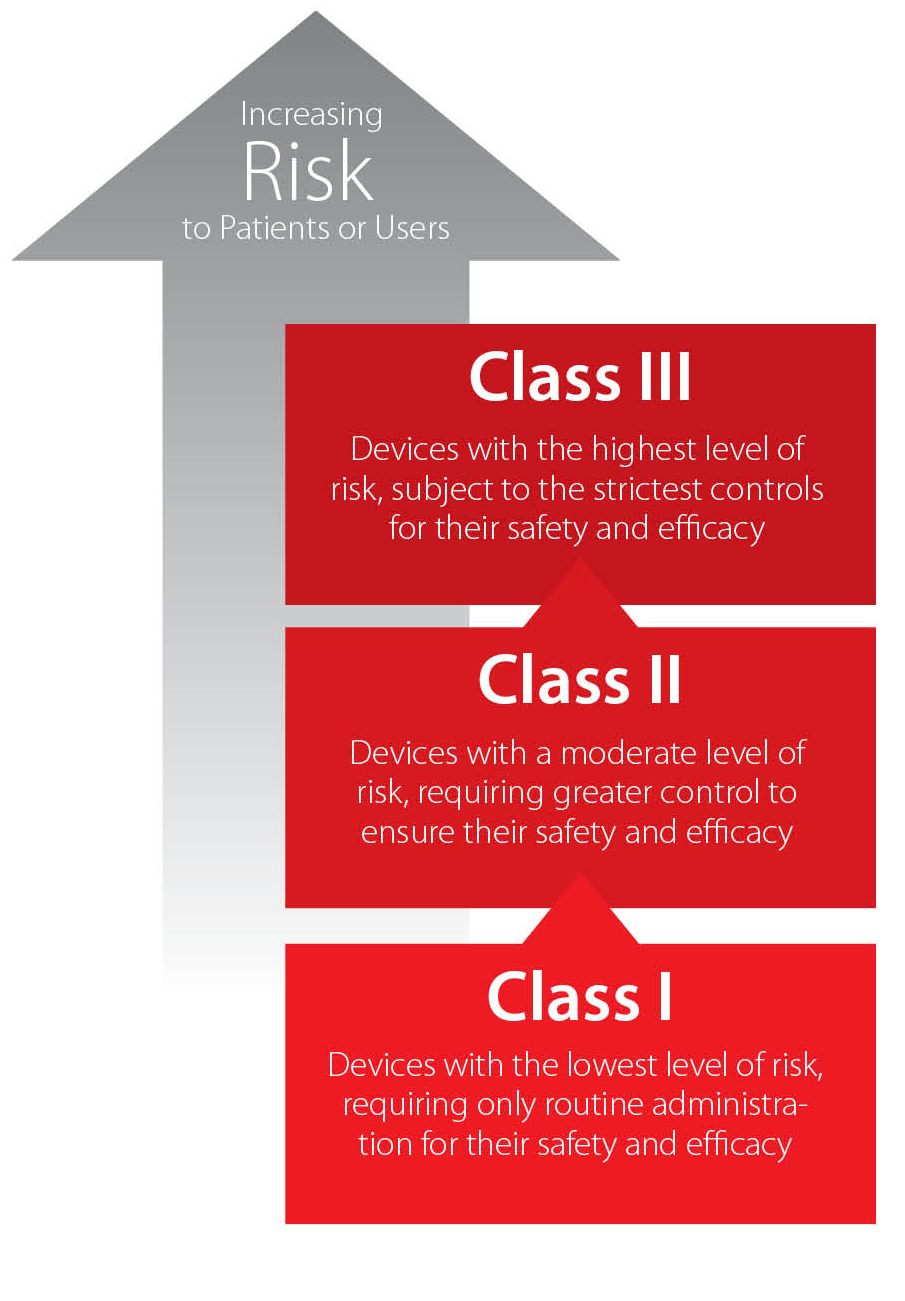
Risk-based classification and administration
The classification regime for medical devices in China differs significantly from those in the European Union and United States. For example, a device considered Class II in the U.S. or Class II-a/II-b in the E.U. may be considered Class III in China, meaning its registration process will be longer and more costly.
Basically, medical device classification in China is determined according to CFDA Order No. 15 (under revision) and other CFDA documents. While there are many factors related to classification, essentially devices are divided into three classes based on the risk they present to patients or users.
Summary
Investing in China’s medical device industry is expected to be a major gain for foreign investors over the coming decades as China’s population ages. The current number of elderly Chinese is estimated to be some 194 million, and this will rise to 300 million by 2025. Running concurrently with this is the fact that China’s social security system as concerns retirement pensions is modest. The implications of this are that China will be calling on its working population to practice traditional “family piety” as a Chinese national characteristic and support the old and infirm.
As China’s middle class is set to rise to some 600 million by 2025, the message is clear: the need for specialist healthcare and lifestyle equipment for the elderly will boom, and will be spearheaded by middle-class consumers. Dealing with investments in the medical device industry however is not so straight forward. Industry and product specific licences have to be obtained, and the procedure is more complex than a straightforward foreign investment. Interested investors are recommended to discuss the regulatory and market entry complexities with China business consultants familiar with the industry.
 This article is an excerpt from the November issue of China Briefing Magazine, titled “China Investment Roadmap: The Medical Device Industry.” In this issue of China Briefing, we present a roadmap for investing in China’s medical device industry, from initial market research, to establishing a manufacturing or trading company in China, to obtaining the licenses needed to make or distribute your products. With our specialized knowledge and experience in the medical industry, Dezan Shira & Associates can help you to newly establish or grow your operations in China and beyond. This article is an excerpt from the November issue of China Briefing Magazine, titled “China Investment Roadmap: The Medical Device Industry.” In this issue of China Briefing, we present a roadmap for investing in China’s medical device industry, from initial market research, to establishing a manufacturing or trading company in China, to obtaining the licenses needed to make or distribute your products. With our specialized knowledge and experience in the medical industry, Dezan Shira & Associates can help you to newly establish or grow your operations in China and beyond. |
![]()
 China Retail Industry Report 2014
China Retail Industry Report 2014
In this special edition of China Briefing, we provide an overview of the retail industry in China and the procedures for setting up a retail shop, focusing specifically on brick-and-mortar physical retail stores. Further, we have invited our partner Direct HR to offer some insights on the talent landscape in the retail industry, as well as tips for recruiting retail personnel in China.
 Adapting Your China WFOE to Service China’s Consumers
Adapting Your China WFOE to Service China’s Consumers
In this issue of China Briefing Magazine, we look at the challenges posed to manufacturers amidst China’s rising labor costs and stricter environmental regulations. Manufacturing WFOEs in China should adapt by expanding their business scope to include distribution and determine suitable supply chain solutions. In this regard, we will take a look at the opportunities in China’s domestic consumer market and forecast the sectors that are set to boom in the coming years.
 Revisiting the Shanghai Free Trade Zone: A Year of Reforms
Revisiting the Shanghai Free Trade Zone: A Year of Reforms
In this issue of China Briefing, we revisit the Shanghai FTZ and its preferential environment for foreign investment. In the first three articles, we highlight the many changes that have been introduced in the Zone’s first year of operations, including the 2014 Revised Negative List, as well as new measures relating to alternative dispute resolution, cash pooling, and logistics. Lastly, we include a case study of a foreign company successfully utilizing the Shanghai FTZ to access the Outbound Tourism Industry.
- Previous Article Market Overview: The Medical Device Industry in China
- Next Article China Law Trends in 2015